 
He predicted that a total of 112 elements would be discovered. He predicted the atomic numbers of unknown elements. What prediction did Mendeleev make that came true less than 20 years later? a. To make most synthetic elements, scientists use powerful machines called a. 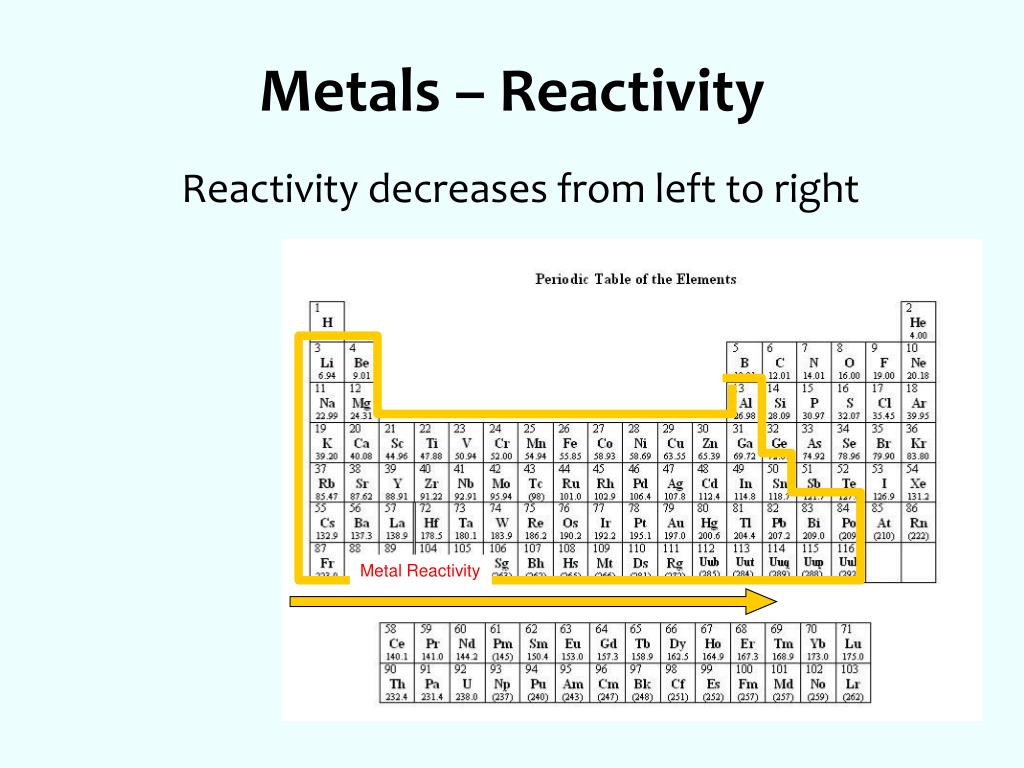
Small nuclei combine to form larger nuclei. Large nuclei combine, then form smaller nuclei. Small nuclei cause large nuclei to break apart. All the nuclei repel each other because of their positive charges. How does nuclear fusion create new elements inside stars? a. The two most common alkaline earth metals are a. The elements have the same atomic number. The elements have a wide range of properties. Which of these statements about a column of the periodic table is true? a. The atomic number of an element is based on the a. have properties that change in a pattern. have the same or nearly the same properties. The elements in a row of the periodic table a. What natural event provides enough energy for the nuclear fusion reactions that create the heaviest elements? a. Which parts of the atom move around the nucleus? a. Which particles in atoms have a negative electric charge? a. Mendeleev created the first periodic table by arranging elements in order of a. At room temperature, more than half of the nonmetal elements are a. in Period 1, the first row across the top. 
in Group 1, the first column on the left. In the periodic table, the most reactive metals are found a. Please use capital letters: A, B, C, or D. 1 Name: Class: Date: Chemical Building Blocks: Chapter 3: Elements and Periodic Table Study Guide Multiple Choice Identify the letter of the choice that best completes the statement or answers the question. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
